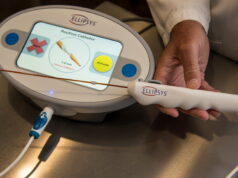
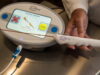
Simergent has announced 510(k) clearance from the US Food and Drug Administration (FDA) for the Archimedes automated peritoneal dialysis (APD) system for the treatment of patients with end-stage kidney disease (ESKD) in clinical and home settings.
Simergent has announced 510(k) clearance from the US Food and Drug Administration (FDA) for the Archimedes automated peritoneal dialysis (APD) system for the treatment of patients with end-stage kidney disease (ESKD) in clinical and home settings.
“Innovative, patient-centric technologies like Archimedes that simplify care, reduce patient burden, and enhance the home dialysis experience have the potential to transform kidney care and enable more patients to successfully adopt home therapies. Congratulations to the Simergent team on receiving FDA 510(k) clearance for the Archimedes automated peritoneal dialysis cycler,” stated Jeffrey Perl (St Michael’s Hospital, Toronto, Canada), editor-in-chief of the Peritoneal Dialysis International journal.
Archimedes is a home dialysis system designed for patient and clinic users with a wide variety of educational and socioeconomic backgrounds, Simergent says in a press release. Its four-hour battery life and integrated mobile cart allows in-home mobility while performing dialysis. The patented fluid delivery and measurement technology requires no pump, which supports quiet operation for this nocturnal therapy.
Archimedes incorporates a patented shroud to support aseptic tubing/bag connections.
“At Simergent, we believe that patients should be allowed to perform dialysis in the comfort of their own homes and not be limited to daytime treatment at a dialysis clinic nor limited to treating in their bedroom at home. Our Archimedes system enables in-home mobility to add some normality to patients’ lives, helping reduce the burden of dialysis care,” commented Steve Lindo, chief executive officer and co-founder of Simergent. “This device gives clinicians a much-needed tool in their toolkit to offer patients a more flexible treatment option with reduced drain suction pressures compared to traditional devices. In addition, it offers hospitals an option to treat patients using their own nursing staff due to our intuitive guided user interface and simple disposable tubing setup. The FDA’s clearance validates our approach to patient-centric kidney care.”
Archimedes will launch in the USA in 2026.