Preliminary clinical results from a first-in-human study assessing the Amplifi vein dilation system (Artio Medical) were presented at this year’s Vascular InterVentional Advances conference (VIVA; 4–7 October 2021, Las Vegas, USA). Data were presented by Surendra Shenoy—an associate professor of surgery at the Washington University School of Medicine at Barnes-Jewish Hospital in St Louis, USA—during the event’s final late-breaking clinical trial session.
Preliminary clinical results from a first-in-human study assessing the Amplifi vein dilation system (Artio Medical) were presented at this year’s Vascular InterVentional Advances conference (VIVA; 4–7 October 2021, Las Vegas, USA). Data were presented by Surendra Shenoy—an associate professor of surgery at the Washington University School of Medicine at Barnes-Jewish Hospital in St Louis, USA—during the event’s final late-breaking clinical trial session.
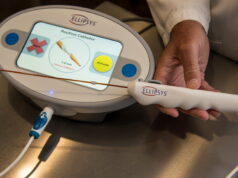
The Amplifi system is designed for percutaneous placement and up to 14 days of use to stimulate arm vein enlargement in haemodialysis patients using rapid, non-pulsatile, venous blood flow. The device includes a wearable, external blood pump, inflow and outflow catheters, and a controller, all of which are removed completely during arteriovenous fistula (AVF) creation. It aims to make more patients eligible for AVF surgery, reduce the time required for AVF maturation and increase successful maturation after AVF creation.
Data presented at VIVA came from the first five patients treated in a prospective, non-randomised, single-arm, first-in-human clinical study with the Amplifi system, and demonstrated that forearm and upper arm mean cephalic vein diameters had more than doubled over an average treatment period of 8.6 days.
AVFs were successfully created using treated veins in all patients. Maturation data are available for the first three patients and show that both the forearm and upper arm AVFs matured quickly with mean outflow vein diameters of 7mm and a mean blood flow rate of more than 1,000 ml/min after a six-week maturation period. No device-related or procedural adverse events were observed.
“Not long after AVFs were first pioneered for haemodialysis in 1966 by doctors Cimino, Brescia, and Appel, concerns were raised about patients who were not suitable for AVF surgery and the high rate of AVF maturation failure,” Shenoy said. “Despite these challenges, AVF remains the preferred option for most patients today. The Amplifi system is the first device I have seen that has real potential to address both of these challenges in a major way, to make good on the promise of providing reliable, long-lasting vascular access sites for this unique and vulnerable patient population.”
In a press release, Artio Medical stated it is planning to set up a US clinical trial of the Amplifi vein dilation system in 2022.