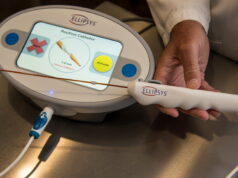
Diality has announced the close of a US$24 million Series B2 investment round. The Series B2 round of funding will be used for activities to support US Food and Drug Administration (FDA) 510(k) clearance of the company’s versatile haemodialysis system—including preparing for a home clinical study and building a US commercial organisation.
“Driven by rising costs and poor outcomes associated with in-centre dialysis, and the rapid shift toward value-based care, dialysis delivery is becoming decentralised. To accommodate cost-effective delivery in all care settings, haemodialysis machines need to be user friendly for non-speciality nurses, patients, and care partners,” said Osman Khawar, CEO at Diality.
“To meet the needs of prescribing physicians and their patients in acute, sub-acute, chronic and home settings, a versatile machine also needs to be capable of delivering a wide range of dialysis prescriptions. We see the lack of a machine with this level of versatility in a compact footprint as a lingering market gap that we seek to fill by bringing to market a best-in-class system capable of delivering the intense dialysis doses commonly prescribed for patients receiving three dialysis treatments per week, in addition to lower-intensity doses suitable for more frequent dialysis.
“The timing of this round positions us to leverage growing interest in this relatively new category of haemodialysis solutions, where we see opportunity for innovation that better addresses the distinct needs of dialysis providers across all segments of the market. Diality is known for innovation, its speed to market and the efficient use of its capital, which will provide significant value to our investors.”
As per a Diality press release, the performance characteristics of this product have not been established. The product is not yet available for use in the EU or USA, and is pending CE-mark approval, and submission and clearance by the FDA, and all performance claims listed are project goals only.