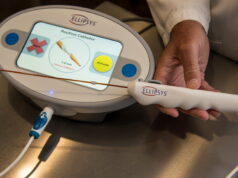
Lydus Medical is pleased to announce that the Vesseal has received US Food and Drug Administration (FDA) 510(k) clearance. The Vesseal is a microvascular anastomosis suture deployment system, for standardised omnivessel anastomoses, enabling simple, fast, safe, and effective procedures.
Lydus Medical is pleased to announce that the Vesseal has received US Food and Drug Administration (FDA) 510(k) clearance. The Vesseal is a microvascular anastomosis suture deployment system, for standardised omnivessel anastomoses, enabling simple, fast, safe, and effective procedures.
An anastomosis is one of the most complicated steps of microvascular surgery and fundamental to the success of these demanding surgical interventions. Varied clinical fields require microvascular anastomosis, including breast reconstruction; head and neck reconstruction; surgical lymphoedema treatment; and vascular access for haemodialysis.
Until now, microvascular anastomoses have been performed manually. Manual microanastomoses are time- and labour-intensive, require a long learning curve, a unique skill set and great surgical dexterity. The Vesseal is designed to mimic the skill set and dexterity needed to deliver quality patient care in microsurgical anastomoses. It provides, simple, accurate, dependable and consistent results through symmetrical placement of eight microsutures at the anastomosis site.
“FDA clearance of the Vesseal is a significant milestone both for innovation in microvascular anastomoses, and for Lydus Medical. It is common knowledge that there are large clinical unmet needs around microvascular anastomosis,” said Jessica Weiss, CEO of Lydus Medical. “Clinicians who used the Vesseal stated that it shows a significant advantage over the manual anastomosis and provides more consistency in the procedure. We believe that the Vesseal will support enhanced outcomes in microsurgical procedures, as well as improved patient care.”