Following a successful proof-of-concept study, Gerard S Goh and Markus P Schlaich have embarked upon a first-in-human study to explore the safety and efficacy of combined renal and common hepatic artery denervation in patients with type 2 diabetes and hypertension. Here, the co-principal investigators describe the rationale behind their single blinded, multi-centre, prospective feasibility study—named the MODUS trial—and explore the “pandemic” of cardiometabolic disease.
Cardiometabolic disease has evolved into a pandemic, resulting in substantial loss in quality of life and enormous economic burden on healthcare systems world-wide. Cardiovascular and metabolic regulation is governed by neural signaling from the sympathetic nervous system in critical organs including the heart, kidneys, liver, pancreas, skeletal muscle, and adipose tissue. Increased sympathetic activation is a hallmark of cardiometabolic disease and has been demonstrated in obesity, metabolic syndrome, type 2 diabetes, hypertension, heart failure, chronic kidney disease, and many other relevant conditions. Insulin resistance, non-alcoholic fatty liver disease (NAFLD), and a systemic pro-inflammatory state are key mediators of the adverse consequences associated with cardiometabolic disease. A self-perpetuating cycle develops where sympathetic overdrive contributes to insulin resistance and subclinical inflammation, which in turn maintain increased sympathetic signaling.

Direct therapeutic targeting of the sympathetic nervous system therefore appears as a rational approach to improve both cardiovascular and metabolic control. Indeed, catheter-based renal denervation using either radiofrequency energy or therapeutic ultrasound has now been shown in four sham-controlled trials to be safe and to effectively lower ambulatory blood pressure in patients both treated and untreated with antihypertensive medications (SPYRAL OFF-MED, SPYRAL ON-MED, RADIANCE-HTN SOLO, RADIANCE-HTN TRIO). Additional salutary effects on glucose metabolism have been reported in some renal denervation studies, suggesting that the beneficial effects of selectively targeting the sympathetic nerves may not be limited to the management of cardiovascular disorders, but may also help to restore metabolic homeostasis. This approach would perhaps be most effective if the sympathetic drive to metabolically-active organs such as the liver (gluconeogenesis), pancreas (insulin release), and duodenum could be inhibited directly.
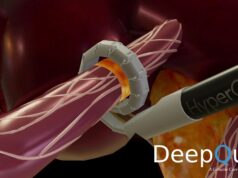
A recent preclinical proof-of-concept study in a porcine model where percutaneous denervation in both renal arteries and the common hepatic artery (CHA) was performed with the iRF Denervation System demonstrated that denervation of both the kidneys and the liver could safely be achieved with reductions in tissue norepinephrine concentration when compared to control animals of 78% and 88% (p<0.001 for both), respectively. Pancreatic and duodenal norepinephrine concentration was also reduced significantly.
Consequently, a first-in-human study has recently been initiated to explore the safety and efficacy of combined renal and common hepatic artery denervation in patients with type 2 diabetes and hypertension.

The MODUS trial (Multi-organ denervation to reduce sympathetic drive, a single blinded, multi-centre, prospective feasibility study) is a multicentre randomised trial that is examining the effects of hepatic and/or renal denervation in patients with hypertension, diabetes, and metabolic syndrome. Patients on one or two antihypertensive and oral anti-diabetic drugs within certain BP and HbA1c ranges undergo a ‘wash-out’ phase of four weeks with regular BP and glucose monitoring before receiving a single session denervation therapy. The trial utilises the iRF denervation system (Metavention), which is a radiofrequency balloon-based catheter with internal cooling where the catheter is inserted into the renal and/or common hepatic arteries and denervation performed. Centres in Perth, Melbourne, and Sydney (Australia) are participating in the trial, and the first two patients were recently enrolled at the Alfred Hospital, Melbourne, treated by Gerard Goh and Warren Clements.
Gerard S Goh is head of Interventional Radiology at The Alfred Hospital, Melbourne Australia, and is adjunct associate professor in the department of Surgery at the Monash University, Melbourne, Australia. He is also president of the Interventional Radiology Society of Australasia (IRSA).
Markus P Schlaich is the Dobney chair in Clinical Research and a nephrologist and hypertension specialist at the School of Medicine—Royal Perth Hospital Unit and the University of Western Australia, Perth, Australia.
Disclosures: Gerard S Goh and Markus P Schlaich are co-principal investigators of the MODUS trial, and as such receive research funding from Metavention.