 July was a month of acute kidney injury (AKI) updates and regulatory shifts, with empagliflozin winning European Medicines Agency approval and the US Food and Drug Administration (FDA) “lowering the red flag” on paclitaxel.
July was a month of acute kidney injury (AKI) updates and regulatory shifts, with empagliflozin winning European Medicines Agency approval and the US Food and Drug Administration (FDA) “lowering the red flag” on paclitaxel.
The European Society for Vascular Surgery (ESVS) guidelines on vascular access recommend minimal diameters of 2mm in the forearm and 3mm in the upper arm in vessels selected for the creation of an arteriovenous fistula (AVF). Whether this recommendation leads to better AVF-related outcomes, however, has been called into question in new research.
The Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) granted a positive opinion recommending Jardiance (empagliflozin) for the treatment of adults with chronic kidney disease (CKD), Boehringer Ingelheim and Eli Lilly and Company announced.
AKI is considered by many to cause accelerated loss of kidney function—but, according to the authors of new research published in the Annals of Internal Medicine, changes in research focus, practice patterns, and public health targets that have taken place in line with this view may have been based on studies with several methodological limitations.
Makana Therapeutics announced that it has been awarded an Artificial Kidney Phase II Prize from KidneyX to continue development of its genetically engineered donor pigs for use in kidney transplantation.
A campaign to reshape kidney care within the National Health Service (NHS) in England was the subject of a centrepiece session at the recent UK Kidney Week event in Wales (5–7 June, Newport, UK), with a host of speakers setting out the ambitions of the Renal Services Transformation Programme (RSTP).
In a letter to healthcare providers dated 11 July 2023, the US FDA communicates that the risk of mortality associated with paclitaxel-coated devices to treat peripheral arterial disease (PAD) is no longer supported based on data and analyses.
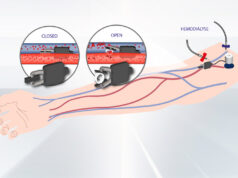
Conventional haemodialysis (CHD) consists of three four-hour sessions a week, but a more regular daily haemodialysis (DHD) regimen of six three-hour sessions a week can improve cardiovascular health in CKD patients. This is according to recent research by Steven G Achinger (University of South Florida, Tampa, USA) and Juan Carlos Ayus (University of California Irvine, Irvine, USA).
Researchers behind a new study published in the Annals of Vascular Surgery, among them lead author Laura Anderson and corresponding author Benjamin Brooke (both University of Utah Health, Salt Lake City, USA), have found that severe obesity risks a greater likelihood of maturation failure in the AVFs of patients looking to start dialysis.
Based on findings from a new study published in the journal The Lancet Regional Health—Americas, researchers at Johns Hopkins Medicine and three collaborating medical institutions suggest that people living with the human immunodeficiency virus (HIV) who donate a kidney to other people living with HIV (PLWH) have a low risk of developing end-stage kidney disease (ESKD) or other kidney problems in the years following the donation.
Patients who engage in light exercise while undergoing dialysis are physically fitter and are admitted to hospital less frequently than those who do not. These are the findings of a large-scale study conducted by a consortium led by the Technical University of Munich (TUM; Munich, Germany). The researchers believe that exercise programs should be offered to dialysis patients as standard.