SyMap Medical has announced that it has completed enrolment of the SMART study, which will evaluate the safety and efficacy of targeted renal denervation (RDN) using the SyMapCath I system for the treatment of uncontrolled hypertension.
SyMap Medical has announced that it has completed enrolment of the SMART study, which will evaluate the safety and efficacy of targeted renal denervation (RDN) using the SyMapCath I system for the treatment of uncontrolled hypertension.
According to SyMap, this is the first pivotal study for registration purpose, worldwide, using targeted RDN technology to treat uncontrolled hypertension.
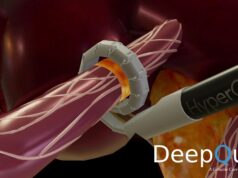
The SyMapCath I system is designed to map renal nerves and provide real-time feedback before, during, and after RDN procedures in order to achieve selective ablation of renal sympathetic nerves.
The SMART study, which has enrolled a total of 220 patients at 16 hospitals in China, is a prospective, multicentre, single-blind, randomised, sham-controlled trial intended to evaluate the safety and efficacy of targeted renal sympathetic denervation using SyMapCath I and SymPioneer in people who have had essential hypertension for at least six months, and in whom blood pressure still cannot be controlled following pharmacotherapy.
The dual primary endpoints for the study are control rate of office systolic blood pressure (SBP) at six months (SBP<140mmHg); and change in composite index based on the classes and doses of antihypertensive drugs to reach the control of office SBP (<140mmHg) at six months.