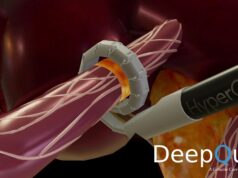
SoniVie recently announced that on 5 May 2022 the US Food and Drug Administration (FDA) granted investigational device exemption (IDE) approval for its REDUCED1 pilot study to treat resistant hypertension patients with renal artery denervation using the company’s therapeutic ultrasound ablation system (TIVUS).
SoniVie recently announced that on 5 May 2022 the US Food and Drug Administration (FDA) granted investigational device exemption (IDE) approval for its REDUCED1 pilot study to treat resistant hypertension patients with renal artery denervation using the company’s therapeutic ultrasound ablation system (TIVUS).
“We are very pleased that the FDA has approved the REDUCED1 (Renal denervation using ultrasonic catheter emitted energy) study. Sites initiation has started, and many clinical teams have responded very favourably about participating in the study. There is a significant number of patients that may benefit from our technology and we are happy to announce this important step towards the introduction of TIVUS in the USA. There is a lack of effective therapeutic solutions for patients suffering from resistant hypertension, and physicians are looking forward to a safe, effective and easy to use device treatment,” said Christian Spaulding, CMO, SoniVie.
“This is a significant US regulatory milestone for SoniVie, starting the feasibility study using the ultrasound ablation platform in the USA for the renal denervation indication. This is a major step and priority in the company’s history,” said Tomaso Zambelli, CEO, SoniVie.
The REDUCED1 study will further expand the company’s clinical experience based on two clinical trials in renal denervation performed using the earlier generation of the TIVUS.
According to SoniVie, renal denervation with TIVUS is a minimally invasive procedure that uses high-frequency non-focused ultrasound energy to ablate nerves in the renal artery. The company claims that this causes a reduction in the nerve activity, which may decrease blood pressure. This procedure is designed for patients who suffer from resistant hypertension.